Chemistry is the analysis of the compounds that form matter from. Chemistry is heavily experimental because you can only study reductions if the substances totally mix together. The term chemistry was used for the first time around A.D. 400-409, and it was used in reference to changing matter. Chemistry is the science that studies atoms and molecules along with their properties. Matter consists of atoms and molecules, so chemistry is all-encompassing and is considered fundamental science.
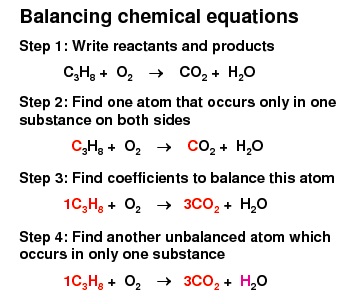
The shortest method of expressing a chemical reaction with the help of formulae is called a chemical equation. A chemical equation can be balanced by balancing the number of various atoms on both sides called balancing chemical equations. Formulas of compounds are never changed to equalise various atoms. A chemical equation is said to be balanced if all the atoms present in the reactants appear in the same numbers among the products.
Basics of Chemistry
Chemistry is often called the central science. It derives this name because of its importance to all the other sciences. Although chemistry did not exist as a modern science until two hundred years ago, humans have used chemistry from prehistoric times. Proof of early chemical applications includes cave paintings dating back to 25000 B.C. It is not difficult to picture our ancestors acquiring natural pigments by pressing berries or combining broken rocks with water to create various colors which can be used as paints. The chemistry definition is “chemistry is the study of the composition, structure and properties of substance and the changes that they undergo“.
- Chemistry is important to us, and in our everyday lives, chemistry takes place all around us.
- Chemistry is the discipline concerned with the universe’s materials and the modifications these elements experience.
A knowledge of chemistry is useful to almost everyone – chemistry occurs all around us all of the time and an understanding of chemistry is useful. Chemistry can also be defined in terms of what chemists do. For example, synthetic chemists combine different types of materials to create new ones that have interesting properties or practical uses.
What is a Balanced Equation?
A chemical equation is balanced because both sides of the equation have the same number of atoms of each form. That means if we have 12 hydrogen on the left side of the equation, we have to have 12 hydrogen on the right side, because if there are 4 oxygen on the left side, 4 oxygen on the right side and so on. This is because of the law of mass conservation-during a chemical reaction we can not create or kill atoms. But we can not only add atoms to each side at random. We have to deal with the reactant molecules. A chemical equation with chemical symbols is also much simpler to use, so then we would be able to see how many atoms of each form are in each element.
Chemistry is often called fundamental research, and with good cause. Many of the anomalies that exist around us in the universe include chemical changes , changes in which one or more chemicals transform into separate chemicals.